Studies
A Retrospective Review of IUI Clinical Outcomes Following Semen Collection in the ProteX versus a Standard Specimen Cup
Preliminary Clinical Outcomes in an IVF Program using the ProteX versus a Standard Specimen Cup for Semen Collection
Early Fertility Trials of Semen Collection Device Previously Demonstrated to Improve Semen Parameters and Pregnancy Rates in Animal Models
Pregnancy Trials Using the Device for Improved Semen Collection
Physiological and Biochemical Assessment of a New Semen Collection Device
Continued Evaluation of a New Semen Collection Technique / Container in Subfertile and Infertile Individuals Using a Cross-species Model
New Semen Collection Technique / Container Improves Semen Quality
Improving Semen Quality Using a Modified Collection Technique
A Novel Collection Technique for Improved Semen Quality
Improvement of the Semen Collection Environment Using a New Semen Collection Device
ProteX Original Research | Human + Animal Study
Continued Evaluation of a New Semen Collection Technique / Container in Subfertile and Infertile Individuals Using a Cross-species Model
Authors
- Department of Animal and Food Sciences, Texas Tech University, Lubbock, TX
- Department of Obstetrics and Gynecology, Texas Tech University Health Sciences Center, Lubbock, TX
Publication
Objective
To date, the common thread in the use of semen collection / extension techniques, whether being used for intrauterine insemination, cryopreserved semen, or advanced reproductive techniques such as in vitro fertilization, is that the samples are collected into a dry, unprotected specimen container. This container, by its nature, allows for drastic shifts in temperature and pH resulting in damage to the spermatozoa.
Preliminary studies from this laboratory using a new collection container / technique to collect semen from canines and humans demonstrated an improvement in most semen parameters. The modified collection device optimizes semen parameters by controlling temperature, pH, and osmotic stress. The objective of the current study was to further investigate the usefulness of the modified device in improving semen parameters using a cross-species model focusing on subfertile and infertile individuals.
Materials and Methods
Human donors were collected into both devices in sequential samples at three-day intervals. Standard semen parameters (viability, motility, morphology, concentration) and biochemical markers were evaluated for all donors at 0, 1, 3, 6, 12, 18, and 24 hours post-processing. Canine and Equine samples were further evaluated at 24-hour intervals until the samples had reached 0% motility. All donors were then classified as either fertile or subfertile based upon species standard for count and / or motility. Data analysis within species were performed with SPSS using the general linear model and appropriate t-tests.
Results
Conclusions
Modification of the semen collection / extension procedure resulted in improved semen parameters for extended time periods post-collection. The data suggests the described technique can yield significantly more motile sperm by placing the sample into a physiologically favorable environment (eliminating pH and cold shock), thus making more sperm available for use in a variety of infertility treatments. Further studies, evaluating pregnancy rates, will be needed to confirm these observations.
Figure 1
Motility Using the DISC – Cross-species view at 24 hours
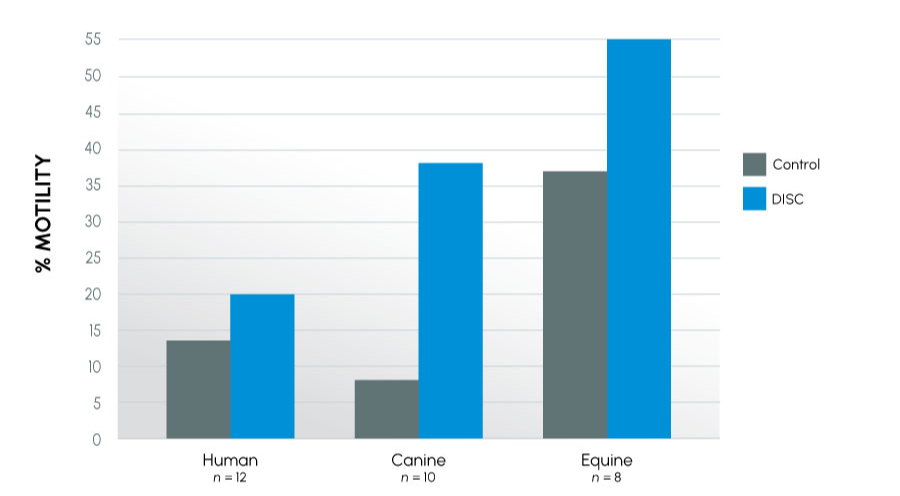
Insights
Figure 2
Time to last full insemination dose – subfertile canine and equine
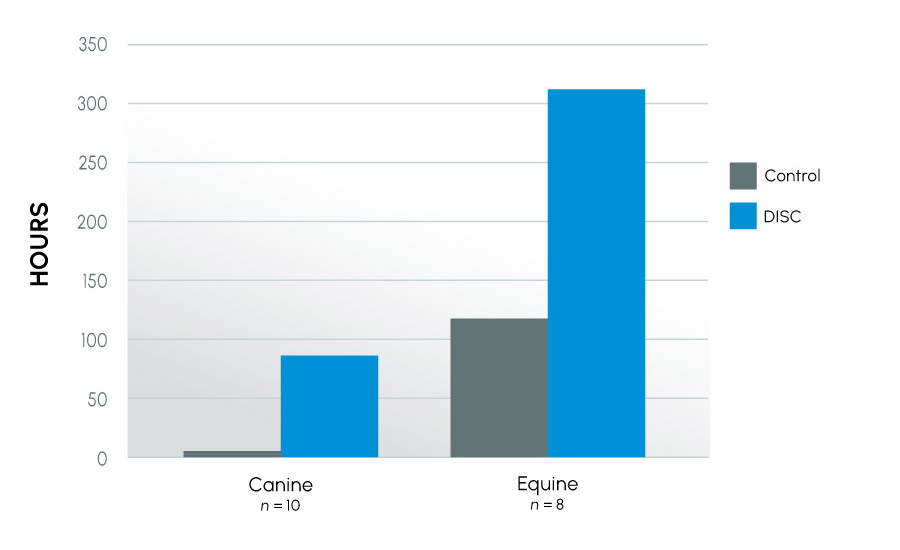
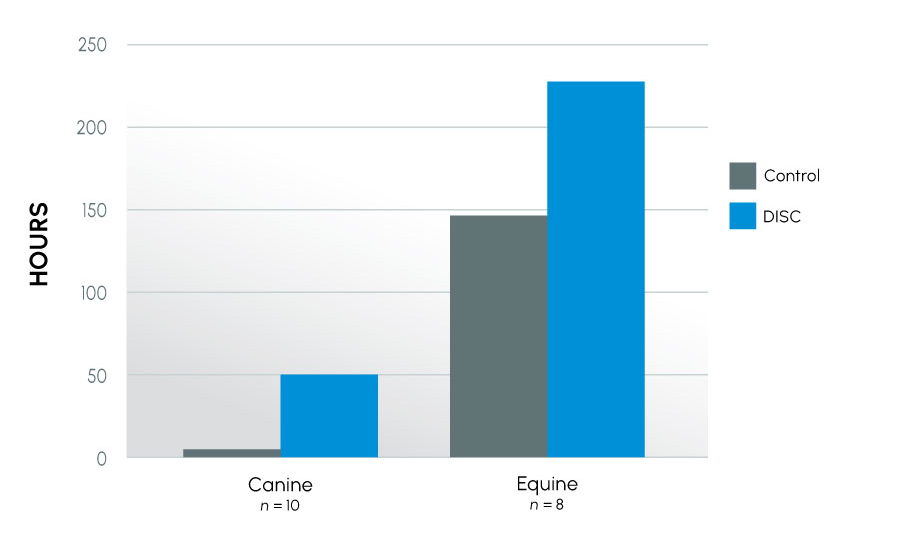
Figure 3
Time to last full insemination dose – all canine and equine
Insights
This series of experiments examined how semen from different species reacted in ProteX. The species evaluated included human, equine, and canine, all species that can have infertility. Both fertile and infertile canine and equine subjects were used, as well as subfertile human subjects. Motility was improved in all species, further, time to last full insemination dose was significantly improved in both canine and equine models. The greatest improvement was seen in subfertile animals. This indicates that protecting the sperm from damage at collection has the greatest impact on the infertile male.
Direct insights into the research, methodology, and results have been added to this summary by the co-inventors themselves. This additional information is intended to provide helpful context to professional practitioners and does not fundamentally change the outcomes or interpretation of the published results. All ProteX research content and material is the property of Reproductive Solutions and may not be redistributed or republished without our consent. All rights reserved.
